 
Also, the starting materials in these cases are different. The ability to correlate these energy differences is justified by the Hammond postulate and the Bell–Evans–Polanyi principle. The p K aH, however, represents the difference in energy between starting materials and products (Δ G°) with differences in acidity reflected in changes in this quantity (ΔΔ G°). Leaving group ability represents the difference in energy between starting materials and a transition state (Δ G ‡) and differences in leaving group ability are reflected in changes in this quantity (ΔΔ G ‡). The correlation between p K aH and leaving group ability, however, is not perfect. 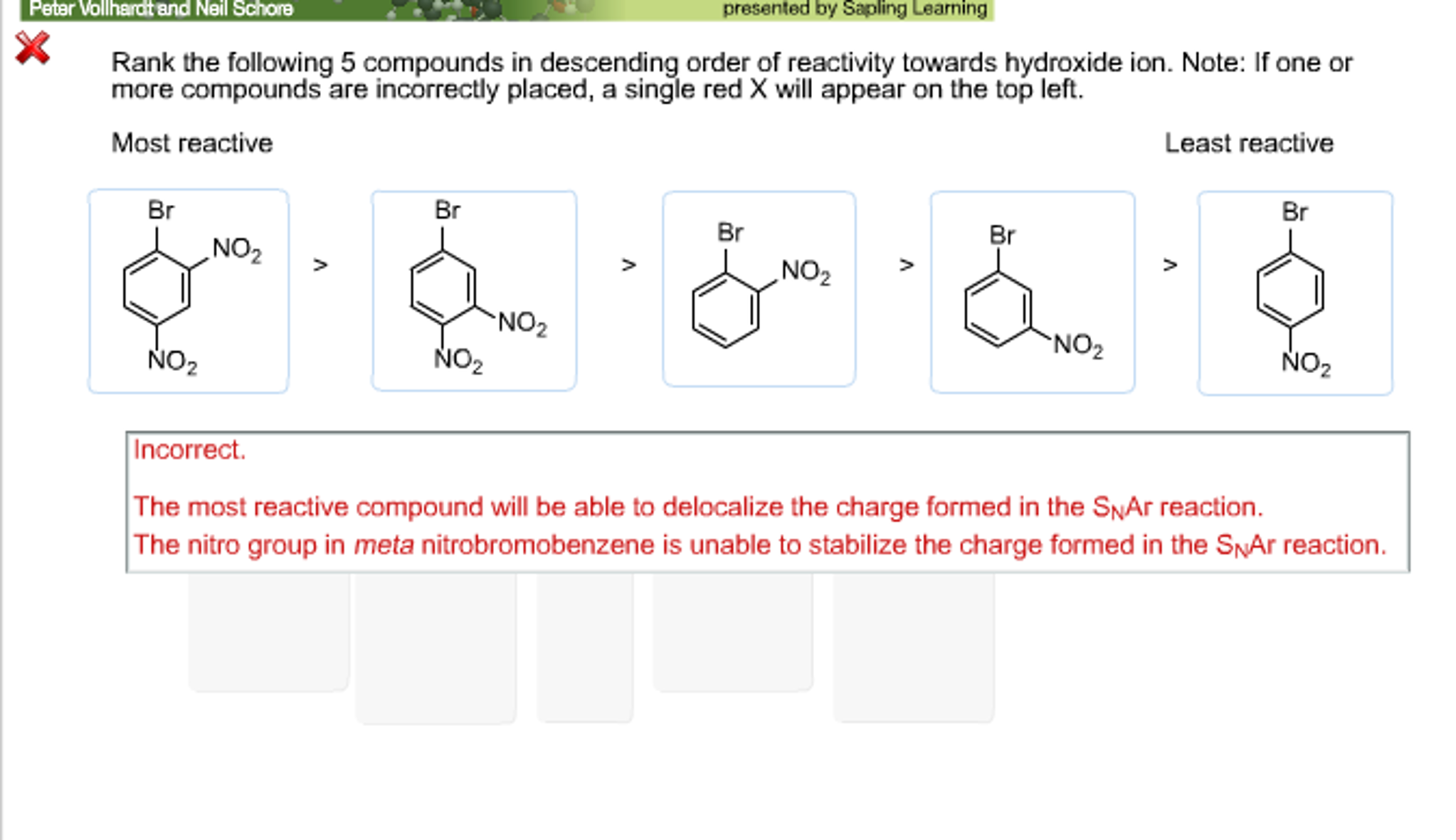
In an ionization reaction, as in all reactions that involve leaving group departure, the leaving group bears a larger negative charge in the transition state and products than it does in the starting materials A good measure of anion stability is the p K a of an anion's conjugate acid (p K aH), and leaving group ability indeed generally follows this trend, with a lower p K aH correlating well with better leaving group ability. Because the leaving group bears a larger negative charge in the transition state (and products) than in the starting material, a good leaving group must be able to stabilize this negative charge, i.e. It is helpful to consider the concept of leaving group ability in the case of the first step of an S N1/E1 reaction with an anionic leaving group (ionization), while keeping in mind that this concept can be generalized to all reactions that involve leaving groups. By transition state theory, this implies that reactions involving good leaving groups have low activation barriers leading to relatively stable transition states. The physical manifestation of leaving group ability is the rate at which a reaction takes place. Top: S N2 reaction middle/left: first step of S N1 and E1 reactions middle/right: second step of E1cb, A AC2, and B AC2 reactions bottom: E2 reaction. In this article, the discussions below mainly pertain to leaving groups that act as nucleofuges.Ĭommon mechanistic contexts that involve the departure of a nucleofugal leaving group. A relatively uncommon term that serves as the antonym of leaving group is entering group (i.e., a species that reacts with and forms a bond with a substrate or a substrate-derived intermediate). 
Similarly, species of high thermodynamic stability like nitrogen ( N 2) or carbon dioxide ( CO 2) commonly act as leaving groups in homolytic bond cleavage reactions of radical species. In the broader IUPAC definition, the term also includes groups that depart without an electron pair in a heterolytic cleavage (groups specifically known as an electrofuges), like H + or SiR + 3, which commonly depart in electrophilic aromatic substitution reactions. Common anionic leaving groups are halides such as Cl −, Br − and I −, and sulfonate esters such as tosylate ( TsO −), while water ( H 2O), alcohols ( R−OH), and amines ( R 3N) are common neutral leaving groups. Ī species' ability to serve as a leaving group depends on its ability to stabilize the additional electron density that results from bond heterolysis. In this context, leaving groups are generally anions or neutral species, departing from neutral or cationic substrates, respectively, though in rare cases, cations leaving from a dicationic substrate are also known. In this usage, a leaving group is a less formal but more commonly used synonym of the term nucleofuge. However, in common usage, the term is often limited to a fragment that departs with a pair of electrons in heterolytic bond cleavage. 
In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. Atom(s) which detach from the substrate during a chemical reaction 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
